Product Description
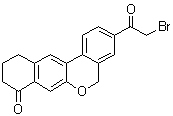
Velpatasvir intermediate 3-(2-Bromoacetyl)-10,11-dihydro-5H-benzo[d]naphtho[2,3-b]pyran-8(9H)-one CAS number is 1378390-29-4, The main methods of synthesis are as follows:
1.1-Bromo-2-bromomethyl-4-chlorobenzene is catalyzed by palladium pivalate, coupled with tri-(4-fluorophenyl)phosphine, coupled to form a 3-chloro-10,11 -Dihydro-5H-benzo[d]naphtho[2,3-b]pyran-8(9H)-one. Then, under the action of palladium acetate catalyzed by Sphos ligand, the ethyl chlorotrifluoroborate is substituted at the position of chlorine to form an olefinic bond, and at the olefinic position, bromine is passed through NBS, and manganese dioxide is oxidized to a carbonyl group to finally obtain Velpatasvir intermediate 3-(2-Bromoacetyl)-10,11-dihydro-5H-benzo[d]naphtho[2,3-b]pyran-8(9H)-one CAS number 1378390-29-4. This method is costly and difficult to control.
2. Using 2-bromo-5-chlorobenzyl bromide and 7-hydroxytetralinone as starting materials, alkylation, intramolecular hydrocarbon activation, ring closure, coupling, reaction with bromine water, oxidation, bromination 3-(2-Bromoacetyl)-10,11-dihydro-5H-dibenzo[C,G]pyran-8(9H)-one CAS 1378390-29-4. The reaction route is long, and the palladium reagent is used twice, which is high in cost.
There is a method to obtain this target compound with reliable quality and low cost. The specific process is as follows:
1.Add 1-[4-bromo-3-(bromomethyl)phenyl]ethanone 29.20 g, 6-bromo-7-hydroxytetralinone 24.11 g, N,N-dimethylformamide 290 mL, stir well, add 27.64 g of potassium carbonate, and react at room temperature for 4-6 hours. At the end of the reaction, add 290 mL of water, 290 mL of ethyl acetate, and separate the liquid. The aqueous layer was extracted once with 145 mL of ethyl acetate. The organic phase was washed twice with water. Dry over anhydrous sodium sulfate, filtered, and concentrated to remove most of ethyl acetate.
2. In the three-necked flask, 45.21 g of yellow powder obtained in the first step, 0.92 g of palladium acetate, 1.68 g of XantPhos, 26.66 g of pinacol borate, 41.46 g of potassium carbonate, and 389 mL of N,N-dimethylacetamide were stirred. After dissolving, the nitrogen gas is switched under vacuum, and the reaction is heated to 60 ° C for 3-4 hours. At the end of the reaction, deoxygenated water is added at 80 ° C to continue the reaction for 4-6 hours, and the mixture is cooled, and the mixture is extracted with 235 mL of isopropyl acetate for 3 times, and the organic phase is washed with water 2 After drying with sodium sulfate, separation by column chromatography and concentration of white powder 22.51 g
3. Add 30.83g of white powder obtained in step 2, 155mL of tetrahydrofuran to the three-necked flask, stir evenly, add 100mL of 20% sodium hydroxide aqueous solution, heat to 50 ° C for 4-6 hours, add dilute hydrochloric acid to adjust the pH to 3~4. Most of the solvent was spun off, cooled to room temperature, filtered, washed with water, and dried in vacuo to give a white powder.
4. To the flask, 29.43 g of the white powder obtained in the step 3 was added, 150 mL of dichloromethane was added thereto, and the mixture was stirred uniformly. 23.79 g of thionyl chloride was carefully added dropwise, and the mixture was refluxed for 3-4 hours after the completion of the dropwise addition. After the completion of the reaction, the methylene chloride was evaporated under reduced pressure to give an oily oil. The oil was added to a mixture of methylene chloride (n), and the mixture was stirred twice, and then dissolved in 150 mL of dichloromethane, and trimethylsilyldiazomethane was added dropwise in an ice water bath. Raise to room temperature for 10-16 hours. At the end of the reaction, the solvent was removed, 150 mL of dichloromethane was added to dissolve, and 21.1 mL of hydrogen bromide acetic acid solution was added dropwise in an ice bath. After the completion of the dropwise addition, the mixture was allowed to react to room temperature for 4-6 hours. After the reaction was completed, 300 mL of saturated sodium hydrogencarbonate was carefully added to quench. The reaction mixture was separated, and the aqueous layer was extracted twice with dichloromethane. The organic phase was washed twice with water, dried over sodium sulfate, and concentrated to remove most solvent, and then petroleum ether (300 mL), beat, and dried by filtration to obtain 30.44 g of the target compound Velpatasvir intermediate 3-(2-Bromoacetyl)-10,11-dihydro-5H-benzo[d]naphtho[2,3-b]pyran-8(9H)-one CAS number 1378390-29-4
Thera. Category: Anti Viral
Cas No.: 1378390-29-4
Synonym: 3-(2-Bromoacetyl)-10,11-dihydro-5H-benzo[d]naphtho[2,3-b]pyran-8(9H)-one 3-(2-Bromoacetyl)-10,11-dihydro-5H-benzo[d]naphtho[2,3-b]pyran-8(9H)-one ;3-(2-Bromoacetyl)-10,11-dihydro-5h-dibenzo[c,g]chromen-8(9h)-on;3-(2-bromoacetyl)-10,11-dihydro-5H-Benzo[d]naphtho[2,3-b]pyran-8(9H)-one;3-(2-Bromo-acetyl)-10,11-dihydro-5H,9H-6-oxa-benzo[a]anthracen-8-one;
Molecular Formula: C19H15BrO3
Molecular Weight:371.22
Assay: ≥99.%
Solubility: Sparingly Soluble (1.2E-3 g/L) (25 ºC), Calc.*
Density: 1.514±0.06 g/cm3 (20 ºC 760 Torr), Calc.*
Packing: Export worthy packing
lMaterial Safety Data Sheet: Available on request
Usage:Velpatasvir Intermediate
Related Intermediate:
1) (2S,5S)-N-Boc-5-Methylpyrrolidine-2-Carboxylic Acid CAS 334769-80-1
2) Ethyl (2S,5S)-5-Methylpyrrolidine-2-Carboxylate CAS 676560-84-2
3) (5S)-N-(Methoxycarbonyl)-L-Valyl-5-Methyl-L-Proline CAS 1335316-40-9
4) 7-Hydroxy-1-Tetralone, MFCD01312225 CAS 22009-38-7
5) (R)-2-(methoxycarbonylamino)-2-phenylacetic acid CAS 50890-96-5
6) 9-Bromo-3-(2-Bromo Acetyl)-10,11-Dihydro-5H-dibenzo(c,g) Chromen-8(9H)-one CAS number is 1438383-89-1
7) 3-(2-bromoacetyl)-10,11-dihydro-5H-Benzo[d]naphtho[2,3-b]pyran-8(9H)-one CAS number 1378390-29-4,
8) GS-5816 interMediate, CAS NUMBER 1378388-16-9

![3-(2-Bromoacetyl)-10,11-Dihydro-5H-Benzo[d]naphtho[2,3-b]Pyran-8(9H)-One For Velpatasvir 1378390-29-4](http://img.nbxc.com/product/1d/f9/dc/16a1feb6b106fc9171946688d9.jpg@4e_360w_360h.src)
.png) Contact Now
Contact Now
