Product Description
Acotiamide hydrochloride trihydrate CAS number is 773092-05-0, marketed in June 2013, which is approved for the treatment of functional dyspepsia (FD), has a very good therapeutic effect on postprandial fullness due to functional dyspepsia, early satiety and Upper abdominal pain . He is the first drug in the world to have proven efficacy and safety in functional dyspepsia patients.it mainly through the inhibition of acetylcholinesterase, promote gastric motility, improve gastric obstruction, enhance gastric dilatation. Since its market launch, Acotiamide hydrochloride hydrate CAS 773092-05-0 has attracted wide attention from the world. Functional dyspepsia is a common gastroenterology disease, some people suffer from the disease as a result of decreased appetite and lower absorption rate, which has led to malnutrition for a long time, and then affect people's quality of life. As people's living standards improve and the deepening understanding of the disease of functional dyspepsia, people with the disease are no longer simply to eat some of the stomach treatment of drugs, they urgently need a dedicated treatment of functional dyspepsia drugs, so The research of Acotiamide hydrochloride hydrate has great value and significance.
The purification and preparation methods of Acotiamide hydrochloride hydrate are described in the literature is as follows:: the crude Acotiamide added to an aqueous solution of inorganic base, after mixing, adding an organic solvent, stirring for crystallization, filtration, and dried to give acotiamide hydrochloride sodium salt or potassium salt, wherein the inorganic base is sodium hydroxide, potassium hydroxide, sodium carbonate or potassium carbonate. The sodium salt or potassium salt of acotiamide hydrochloride is added to the aqueous isopropanol solution, mixed well, acidified with hydrochloric acid to pH = 1-2, stirred for crystallization, heated and dissolved, cooled and crystallized, dried under reduced pressure to obtain Acotiamide hydrochloride hydrate. The disadvantage of this method is that a large amount of organic and inorganic salts are produced during the hydrochloride acid reaction of the inorganic base and crude Acotiamide hydrochloride,and are difficult to remove resulting in a low content of the final product. With the required of Acotiamide hydrochloride hydrate,, multiple repeated recrystallizations are required, which results in low product yields, long production cycles, and high production costs.
There is a simple and feasible method for the preparation of Acotiamide hydrochloride hydrate, prepared as follows:
1) 1 part by weight of crude Acotiamide hydrochloride , 1 to 3 parts by volume of water, 1 to 3 parts by volume of aliphatic alcohol (methanol, ethanol, isopropanol) was added to the reaction flask, the temperature was controlled at 10 ~ 20 ℃, open stirring;
2) fractionally adding 0.95 ~ 1.05 molar parts of sodium bicarbonate;
3) add sodium bicarbonate, continue stirring for 3 to 4 hours;
4) filter, add 1 ~ 2 parts by volume of water to wash the filter cake;
5) wet vacuum drying, controlling the drying temperature of 45 ~ 55 ℃, dried in vacuo to a moisture ≤ 2.0%, to obtain Acotiamide hydrochloride trihydrate CAS number is 773092-05-0
Thera. Category:Antiulcerative,GASTROINTESTINAL DRUGS
Cas No.:773092-05-0
Synonym:Ym-443;AcotiaMide hydrochloride trihydrate;Acotiae;4-Thiazolecarboxamide, N-[2-[bis(1-methylethyl)amino]ethyl]-2-[(2-hydroxy-4,5-dimethoxybenzoyl)amino]-,hydrochloride,hydrate(1:1:3);Acotiamide HCl;Acotiamide hydrochloride hydrate;N-(2-(Diisopropylamino)ethyl)-2-((2-hydroxy-4,5-dimethoxybenzoyl)amino)-1,3-thiazole-4-carboxyamide monohydrochloride trihydrate;Unii-nmw7447A9a;
Molecular Structure: 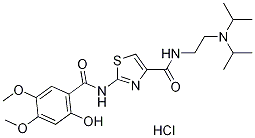
Molecular Formula:C21H31ClN4O5S
Molecular Weight:487.013
Pharmacopeia: In house Spec.
Specifications:Available on request
Packing:Export worthy packing
Material Safety Data Sheet:Available on request

.png) Contact Now
Contact Now
