Product Description
Mizolastine cas number is 108612-45-9 which is a specific and selective peripheral histamine H1 receptor antagonist with anti-histamine and anti-allergic activity. For seasonal and perennial allergic nasal itch, allergic conjunctivitis, urticaria and other allergic symptoms. Mizolastine orally absorbed rapidly, the plasma concentration peak time of 1.5 hours, bioavailability of 65% for the linear metabolic kinetics. The average elimination half-life was 13.0 hours and the plasma protein binding rate was 98.4%. Mizolastine in patients with liver dysfunction absorbed more slowly, with a longer distribution and a moderate increase in AUC (approximately 50%). The main metabolic pathway is the glucuronidation of the parent compound. Cytochrome P4503A4 enzyme system is also involved in the hydrolytic metabolism of this product. The known metabolites have no pharmacological activity. Mizolastine has a unique dual antihistamine and anti-allergic response to inflammatory mediators. In vivo and in vitro pharmacological tests have shown that mizolastine is a potent and highly selective histamine H1 receptor antagonist. Mizolastine cas number 108612-45-9 also inhibits the release of histamine from activated mast cells and suppresses the chemotaxis of inflammatory cells such as neutrophils in an animal model of allergic reaction. At the same time, mizolastine also inhibits the release of intercellular adhesion molecule-1 during allergy. Pharmacological tests showed that mizolastine has anti-inflammatory activity, which may be related to its inhibitory effect on 5-lipoxygenase.
Thera. Category:anti-histamine and anti-allergic
Cas No.: 108612-45-9
Synonym: lndobufen;INDOBNFEN;2-[4-(1-oxo-2-isoindolinyl)phenyl]butyric acid
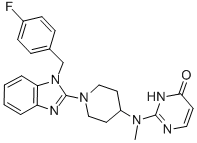
Molecular Weight: 432.49
Molecular formula:C24H25FN6O
Assay: ≥98.%
Appearance: White Crystalline solid
Packing:Export worthy packing
Material Safety Data Sheet:Available on request


.png) Contact Now
Contact Now
