Product Description
Ibudilast cas number is 50847-11-5, it is a colorless plate crystal or white crystalline powder, soluble in organic solvents, slightly soluble in water. Mainly suitable for people over the age of 16 who got asthma, but also improve the sequelae of cerebral infarction, cerebral hemorrhage sequelae and cerebral arteriosclerosis in patients with symptoms. its preparation is mainly capsules, taking the drug after 72 hours, about 60% in the form of metabolites with the urine. Patients disabled who is allergic to the goods, intracranial hemorrhage has not yet fully controlled. Pediatric, pregnant, lactating women disabled. It is different with bronchial expansion drugs and corticosteroids,Ibudilast can not quickly alleviate the symptoms of the attack. Acute cerebral infarction and liver dysfunction in patients with caution. If there are rash, itching and other allergic symptoms, should be discontinued.
Thera. Category: Antiinflammatory
Cas No.: 50847-11-5
Synonym:IBUDILAST;IBUDILAST-D7;KC-404;2-METHYL-1-[2-(1-METHYLETHYL)PYRAZOLO[1,5-A]PYRIDIN-3-YL]-1-PROPANONE;3-ISOBUTYRYL-2-ISOPROPYLPYRAZOLO(1,5-A)PYRIDINE;2-methyl-1-(2-(1-methylethyl)pyrazolo(1,5-a)pyridin-3-yl)-1-propanon;1-PROPANONE, 2-METHYL-1-[2-(1-METHYLETHYL)PYRAZOLO[1,5-A]PYRIDIN-3-YL];2-isopropyl-3-isobutyrylpyrazolo[1,5-a]pyridine;
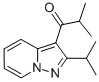
Molecular Formula: C14H18N2O
Molecular Weight: 230.31
Assay: ≥98.%
Packing:Export worthy packing
Material Safety Data Sheet:Available on request
|
Appearance
|
White to off-white crystal or crystalline powder
|
White crystalline powder
|
|
Identification
|
HPLC
|
Conforms
|
|
Solubility
|
Freely soluble in methanol, ethanol and diethyl ether, almost insoluble in water
|
Conforms
|
|
Melting point
|
54.0℃ ~ 57.0℃
|
55.0℃ ~ 56.0℃
|
|
Loss on drying
|
≤0.5%
|
0.20%
|
|
Residue on ignition
|
≤0.1%
|
0.01%
|
|
Heavy metals
|
≤10ppm
|
<10ppm
|
|
Related substances
|
0.1%Single impurity≤0.1%
|
0.03%
|
|
|
Total impurities≤0.5%
|
0.07%
|
|
Assay(on dried basis)
|
≥99.0%
|
99.87%
|
|
Conclusion
|
This product conforms to Enterprise Standard.
|

.png) Contact Now
Contact Now
