Product Description
The first method about Azilsartan N-2 cas 1397836-41-7, use 3-nitro-phthalic acid as the starting material, after esterification, acylation, rearrangement and other steps to obtain the target compound, the route using sodium azide and ethyl chloroformate as the reaction raw materials , These two kinds of raw materials have a certain risk. And this route has long production cycle, low yield, high cost of synthesis. The second,use 2-ethoxybenzimidazole-7-carboxylic acid methyl ester as raw material by substitution, oximation two steps to prepare the target compound, add 2-cyano-4 ' - Bromomethyl biphenyl in the first step of the substitution reaction, this product is expensive, not easy to get. The Third,Methyl 1 - [(2'cyanobiphenyl-4-yl) methyl] -2-ethoxybenzimidazole-7-carboxylate Under the action of hydroxylamine hydrochloride and triethylamine, react in DMSO to obtain the target compound. However, under this condition, not only the reaction time is long, but also amide impurity is produced as much as the target product; the post-treatment method is very complicated; and the actual yield is low.
Considering the huge market prospect of azilsartan, many researchers agree that it is of great industrial value to improve the preparation methods of azilsartan intermediates, improve yield, simplify procedures and reduce costs. Through unremitting efforts, the researchers surprised find that under certain reaction conditions, by controlling the fed into amount of sodium bicarbonate, hydroxylamine hydrochloride and 1 - [(2 'cyanobiphenyl-4-yl) methyl] -2-ethoxybenzimidazole-7-carboxylic acid methyl ester can be conveniently used to prepare the target compound Azilsartan N-2 cas 1397836-41-7 in which the amide impurity can also be significantly reduced. The reaction time is reduced from the original 24 hours to 10 hours, which improves the efficiency. The aftertreatment process only needs to add water to precipitate the product. After simple washing with water and alcohol, the next step can be carried out, which greatly simplifies the refining process.
Thera. Category: Anti-Hypertension
Cas No.:1397836-41-7
Synonym:Ethyl-2-ethoxy-3-((2'-(N'-hydroxycarbaMiMidoyl) biphenyl-4-yl) Methyl)-3H-benzo[d] iMidazole-4-carboxylate;(Z)-Ethyl-2-ethoxy-3-((2'-(N'-hydroxycarbaMiMidoyl) biphenyl-4-yl) Methyl)-3H-benzo[d] iMidazole-4-carboxylate;Azilsartan Impurity 26;2-Ethoxy-1-[[2'-[(hydroxyamino)iminomethyl][1,1'-biphenyl]-4-yl]methyl]-1H-benzimidazole-7-carboxylic acid ethyl ester;ethyl (Z)-2-ethoxy-1-((2'-(N'-hydroxycarbamimidoyl)-[1,1'-biphenyl]-4-yl)methyl)-1H-benzo[d]imidazole-7-carboxylate;
Molecular Structure: 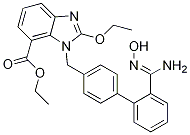
Molecular Formula:C26H26N4O4
Molecular Weight:458.50904
Pharmacopeia: In house Spec.
Specifications:Available on request
Packing:Export worthy packing
Material Safety Data Sheet:Available on request

.png) Contact Now
Contact Now
