Product Description
EPZ-5676 CAS number is 1380288-87-8, a new drug for the treatment of a rare subtype of leukemia EPZ-5676 is recognized by the FDA as a rare disease. The drug is currently in Phase I clinical studies. Affected by this news, the company's stock price also rose.EPZ-5676 CAS number 1380288-87-8 is an inhibitor of an enzyme called DOT1L. DOT1L plays an important role in the pathogenesis of mixed cell line leukemia. Epizyme has authorized Celgene to own the EPZ-5676 sales rights outside the US market.
According to the FDA's rules for rare disease medications, diseases with a patient count of less than 200,000 can be defined as rare diseases, compared with approximately 5,000 patients with mixed cell line leukemia. Epizyme's EPZ-5676 study will receive a series of preferential measures such as tax cuts after qualifying for the rare disease drug.
EP-5676 is a competitive inhibitor of S-adenosylmethionine (SAM) of protein methyltransferase DOT1L with a Ki of 80 pM, which is more than 37,000 more selective than all other PMTs, inhibiting tumor H3K79 methylation. . Phase 1.EPZ-5676 reduced H3K79 dimethylation in MV4-11 cells with an IC50 of 2.6 nM. EPZ-5676 reduces H3K79 methylation in a concentration- and time-dependent manner, but has no effect on other histone sites, which results in a MLL gene and selective apoptotic cell killing of key targets that are inhibited in MLL rearranged leukemia cells. EPZ-5676 inhibited the proliferation of the MLL-AF4 rearranged cell line MV4-11 with an IC50 of 9 nM.
In a MLL rearranged leukemia xenograft model, EPZ-5676 resulted in a dose-dependent anti-tumor activity after a continuous intravenous infusion for 21 days. At the highest dose of 70.5 mg / kg / day, the treatment stopped after the tumor completely resolved, and there was no tumor regrowth for up to 32 days. No significant weight loss or significant toxicity occurred in the EPZ-5676 treated rats.
Thera. Category:Anti-cancer
Cas No.: 1380288-87-8
Synonym:(2R,3R,4S,5R)-2-(6-aMino-9H-purin-9-yl)-5-((((1r,3S)-3-(2-(5-(tert-butyl)-1H-benzo[d]iMidazol-2-yl)ethyl)cyclobutyl)(isopropyl)aMino)Methyl)tetrahydrofuran-3,4-diol
Molecular Formula:C30H42N8O3
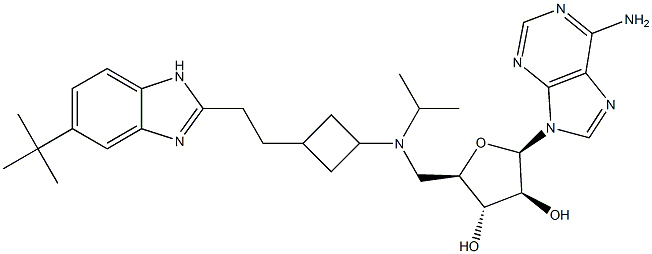
Molecular Weight:562.70628
Pharmacopeia: in house spe.
Specifications:Available on request
Packing:Export worthy packing
Material Safety Data Sheet:Available on request

.png) Contact Now
Contact Now
