Product Description
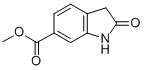
Methyl 2-oxoindole-6-carboxylate CAS 14192-26-8,His synthesis is mainly divided into two steps, the specific operation process is as follows: The first step: 200g (1mol) 4-chloro-3-nitrobenzoic acid suspended in methanol, added 59g (490mmol) Thionyl Chloride within 15min, heated under reflux and reacted for 3 hours at 70℃ and then cooled to 5℃. The product was isolated by centrifugation and dried at 45℃ to give 189.2 g of a solid as a first step, 4-chloro-3-nitrobenzoate ester. Second step: 128.7 g of dimethyl malonate and 107.3 g of sodium tert-pentoxide were added to 350 mL of hot melt solution of N-methyl-2-pyrrolidone; 100 g of 4-chloro-3- Methylbenzoate to 250 mL of the above N-methyl-2-pyrrolidone mixed solution were added at 75℃. then stirring at about 75℃ for 1.5 hours and cooling to 20℃, the mixture was acidified with 1000 mL of dilute hydrochloric acid to pH = 1 and 137.6 g of a solid was filtered off. The solid was dissolved in 880 mL of acetic acid and 10% palladium carbon was added at 45℃to process hydrogenation; after it was stopped, the reaction material was heated to 115℃ for 2 hours. The catalyst was filtered off and 1800 mL of water was added at about 50℃. The reaction product was re-cooled to 5 ℃and centrifuged,dried at 50 ℃ to give 69.6g of a solid, which is the target compound Methyl 2-oxoindole-6-carboxylate CAS 14192-26-8.
3-Oxindole-6-carboxylic acid methyl ester was obtained after the steps of substitution, condensation, deprotection and reduction. In the first step of the substitution reaction, it is necessary to control the substitution temperature of the methyl 2-oxindole-6-carboxylate in stages because of the substitution of 2 positions. The first-stage acetyl protecting group, which has a high substitution temperature and fast reaction, control time can control by-products; the second stage substitution can reduce the temperature, one can ensure that the replacement of the protective group to continue, the second to ensure the condensation of original triethyl benzoate.
Nintedanib intermediate named Methyl 2-oxoindole-6-carboxylate CAS 14192-26-8, it is the nucleus of Nintedanib and is also a versatile intermediate. it can be used to synthesize Nintedanib or Gedatolisib. Compared to the other intermediates of Nintedanib, This intermediate market quantity is the most biggest and we are making commercial quantity for this intermediate too. We can provide 20 grams samples in free of charge, and can provide HPLC,COA AND MSDS, etc for customer reference, after order confirmation, we can provide analytical methods. If customer need presample to confirm quality, we can unconditionally retain the related batch of goods for 20 days. We only have one specification of Methyl 2-oxoindole-6-carboxylate CAS 14192-26-8,different batches do not have significant difference.
Thera. Category: A Potent Kinase Inhibitor
Cas No.: 14192-26-8
Synonym:2-OXO-6-INDOLINECARBOXYLIC ACID METHYL ESTER; METHYL 2-OXINDOLINE-6-CARBOXYLATE;METHYL 2-OXOINDOLINE-6-CARBOXYLATE;METHYL OXINDOLE-6-CARBOXYLATE;OXINDOLE-6-CARBOXYLIC ACID METHYL ESTER
Molecular Formula: C10H9NO3
Molecular Weight: 191.19
Purity: ≥98.%
Packing:Export worthy packing
Material Safety Data Sheet:Available on request
BIBF1120 (Vargatef) is an indolinone derivative potently blocking VEGF-, PDGF-, and FGF-receptor kinases; an indolinone as triple angiokinase inhibitors.
1) Methyl 2-oxoindole-6-carboxylate CAS 14192-26-8
2) Methyl 1-acetyl-2-oxoindoline-6-carboxylate CAS 676326-36-6


.png) Contact Now
Contact Now