Product Description
4-methylcatechol CAS number is 452-86-8, It is a widely used organic compound, can be used for the preparation of pharmaceutical active compounds, It can also be used for the synthesis of antibacterial agents and antioxidants, and can be used as a high-impedance Polymerizers are used for the sterilization and anti-mildew of photosensitive materials and the production of apple-type perfumes. They are used in the daily life industry and have wide application prospects.
There has been extensive research on the synthesis of 4-methylcatechol cas 452-86-8. A one-step catalytic reaction using titanium-silicon molecular sieves and other catalysts has been reported. However, the highest yield is only 12% . Most of the raw materials need to be recycled, and the selectivity of the catalyst is low Or the yield is not high, and recycling is difficult. German experts use hydrogen peroxide for the hydroxylation of phenols in the presence of strong acids. The acid used is sulfuric acid or perchloric acid, which causes serious corrosion to the equipment, and the yield is less than 30%. Another article in Germany reported that sulfur dioxide or selenium dioxide is used as a catalyst to oxidize with almost anhydrous hydrogen peroxide. Although this method does not have strong acid corrosion,However, it is necessary to add an aldehyde compound or a complex component to intensify the process. The operation is cumbersome, and the almost anhydrous hydrogen peroxide solution has a large potential safety hazard. The selenium dioxide used is a highly toxic substance, which easily causes heavy metal pollution. In terms of biology, there are European and American patents that use catechol oxidase to catalyze the synthesis of 4-methylcatechol from p-cresol. Although the yield is high, the reaction conditions are harsh. It is not suitable for industrial production because of the maintenance of the activity of oxidase.
In view of the above-mentioned problems, p-cresol was used as a starting material to produce p-cresol acetate by acylation reaction and Fries rearrangement reaction in high-boiling inert solvent to obtain 2-hydroxy-5-methylacetophenone. Alkalinization into salt, low-temperature oxidation, the reaction is completed, the reaction solution by reduction, acidification, extraction and concentration of crude products, through distillation to obtain pure 4-methyl catechol. The advantage of this method is that the first two reaction products do not need to be purified, and the final product can be directly obtained by one-pot reaction. The operation is simple and the production cycle is short.
Thera. Category: Organic Raw material
Cas No.:452-86-8
Synonym: 4-Methyl-1,2-dihydroxybenzene;4-methyl-2-benzenediol ;4-methyl-benzene-1,2-diol ;4-methyl-pyrocatecho ;p-methylcatechol ;p-Methylpyrocatechol;2-Hydroxy-4-methylphenol;4-Methyl-1,2-benzenediol (4-methylpyrocatechol);
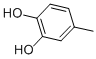
Molecular Formula: C7H8O2
Molecular Weight: 124.14
Specifications: Available on request
Packing: Export worthy packing
Material Safety Data Sheet: Available on request

.png) Contact Now
Contact Now
