Product Description
Roflumilast CAS number is 162401-32-3, Roflumilast is an oral selective phosphodiesterase-4 (PDE4) inhibitor for the treatment of a new type of chronic obstructive pulmonary disease of asthma and chronic obstructive pulmonary disease (COPD) treatment. Roflumilast is mainly expressed in inflammatory cells associated with asthma, including eosinophils, neutrophils, and mast cells. It can specifically act on an enzyme involved in smooth muscle contraction, prevent cAMP degradation, thereby blocking the signaling of pro-inflammatory response, has anti-inflammatory activity, and has a good therapeutic effect in the clinical treatment of asthma and chronic obstructive pulmonary disease. Roflumilast CAS number 162401-32-3 also significantly delays the deterioration of respiratory symptoms and greatly improves the quality of life of patients. It has been shown to inhibit inflammation associated with (COPD) in a new way. As a tablet that is taken orally once a day, roflumilast is not only the first drug in a new treatment for severe COPD, but also the first oral anti-inflammatory drug for patients with COPD. Its unique properties help to better treat patients with chronic obstructive pulmonary disease: when combined with bronchodilators for the treatment of patients with the most severe chronic obstructive pulmonary disease, roflumilast can provide additional reductions in symptoms and disease exacerbations. The benefit is thus the first drug to target a specific phenotype of chronic obstructive pulmonary disease, a patient with a history of severe airflow limitation associated with prolonged cough and phlegm and a history of recurrent disease progression.
Roflumilast is administered orally and is metabolized to N-oxide by cytochrome oxidase P450 (CYP) 3A4 and CYP1A2 enzymes after entering the body. The activity of roflumilast N-oxide is only 2 to 3 times weaker than that of roflumilast, and it also has a high PDE-4 selectivity. About 90% of PDE-4 inhibition in humans is caused by roflus. The other 10% produced by the special N-oxide is produced by the roflumilast prototype. According to FDA-approved roflumilast medication guidelines, there are potential mental health risks, including changes in mood, thinking or behavior, and unexplained weight loss. Roflumilast is not expected to be used for the treatment of other symptoms of COPD, including emphysema. Roflumilast cannot be used for the treatment of sudden respiratory problems and is not recommended for patients under 18 years of age. The main adverse reactions reported include: diarrhea, nausea, headache, insomnia, back pain, loss of appetite and dizziness.
Thera. Category: Anti-inflammatory
Cas No.:162401-32-3
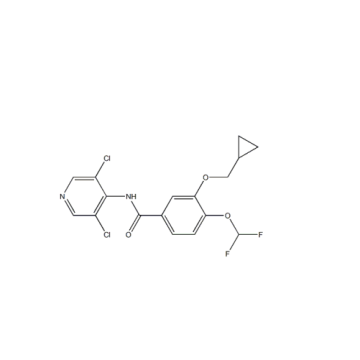
Synonym:3-CyclopropylMethoxy-N-(3,5-dichloro-pyridin-4-yl)-4-isopropoxy-benzaMide;APTA2217;3-Cyclopropylmethoxy-4-difluoromethoxy-N-[3,5-dichloropyrid-4-yl]-benzamide;162401-32-3;Roflumilast intermediates;Roflumilast, >=99%;ROFLUMILAST;3-(Cyclopropylmethoxy)-N-(3,5-dichloro-4-pyridinyl)-4-(difluoromethoxy)benzamide

Molecular Formula:C17H14Cl2F2N2O3
Molecular Weight:403.2074664
Assay: ≥99.%
Packing: Export worthy packing
Material Safety Data Sheet: Available on request

.png) Contact Now
Contact Now
