Product Description
Valbenazine CAS number is 1025504-45-3, development code is NBI-98854,which is a novel, highly selective VMAT2 inhibitor that regulates the release of dopamine during nerve conduction but has no effect on other monoamines, thus reducing Side effects caused by off-target. Valbenazine is capable of maintaining a low sustained concentration of active drug in the plasma and brain, thereby minimizing side effects caused by excessive reduction of monoamine. It is approved by the FDA on April 11, 2017 for the treatment of tardive dyskinesia.
Most of the existing literature does not mention the crystalline form of Valbenazine CAS 1025504-45-3. In the latest study, two crystal forms of Valbenazine were found, namely CS1 and CS2. The two crystal forms are simple and reproducible, and the prepared crystal form has good crystallinity. CS1 is needle-like, CS2 is blocky, and has good dispersibility and few agglomeration. This property is very suitable for the development of pharmaceutical preparations. It has been verified that the solubility and wettability of CS1 and CS2 are in compliance with medicinal requirements and can be stored stably. The most important thing is that these two crystal forms will not undergo crystal transformation during the production process, thus avoiding changes in drug solubility, dissolution rate, bioavailability and efficacy.
Thera. Category:Tardive Dyskinesia,TD
Cas No.:1025504-45-3
Synonym:Valbenazine;NBI-98854
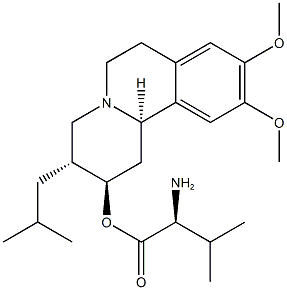
Molecular Weight: 418.56952
Molecular formula: C24H38N2O4
Purity: ≥99.%
Packing:Export worthy packing
Material Safety Data Sheet:Available on request

.png) Contact Now
Contact Now
